Fragment from the Palace of Apries
Summary
The examination and analysis of the painted fragment point in different directions. Part of the chromatic scheme is well attested. Copper-based pigments such as the synthetic Egyptian blue and atacamite were commonly used. Calcium carbonate experienced wide application in time and space from the 5th Dynasty to the Roman period and the use of orpiment is certainly attested from the Middle Kingdom and has been identified on a number of monuments. But the discovery of what appears to be a lead white beneath a layer of orpiment is unusual since the introduction of artificial lead based pigments usually are attributed to the influence of Graeco-Roman tradition. It is possible that the fragment has been repainted since the palace complex was redecorated during the reigns of Apries’ successors which could explain the use of lead white as a second and finer ground. The discrepancies in these preliminary results call for further research.
Description of object
The fragment shows a section of the serekh frame. The frame is carved in slightly raised relief and painted in bright colours. The name of the king, now lost, was written in the upper, yellow part of the frame, while the lower part was decorated with a pattern imitating the façade of a Palace.
Choice of methods
Visual examination
- Macroscopic
- Microscopic in situ
Technical imaging
- UV
- VIL
- IR
Sampling
- XRF
- GC-MS
- ICP MS
Visual examination
The surface is weathered and, in addition to the extensive micro cracks, parts of the original painting have been lost, thus exposing the underlying limestone. Prior to the painting, guidelines were incised with a pointed instrument into the limestone. The preparatory incisions are visible to the naked eye where the paint is lost. The painting of the fragment is divided into two parts: the left half bears only a white ground whereas the right half is painted in green, yellow and red colours on top of a white ground. The green-coloured elements, which are supposed to resemble vegetal growth adorning the palace façade, are usually framed by a bright red outline. Red colour also appears in a somewhat darker and irregular version on parts of both the yellow and green colour. The black paint is limited to the lower section of the fragment forming parts of the green decoration.
Technical imaging
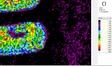
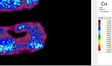
UVL UVL imaging shows two different luminescence phenomena related to the white ground. The white ground which is partly visible beneath the yellow colour appears green whereas the remaining white ground on the rest of the fragment displays a strong blue luminescence. The bright yellow colour emits a greenish luminescence in UV-light. The green-coloured details appear completely dark and are composed of an absorbent and non-fluorescing material. It is not possible to distinguish the bright red outline and the superimposed red layers also appear dark and non-fluorescing on the UVL image. The red colours on the fragment can be affected by the absorbent properties of the green and yellow colours and does not imply the absence of organic materials. VIL The VIL-imaging reveals extensive luminescence properties of Egyptian blue. A regular distribution of relatively uniform grains shining bright white are observed on the main part of the fragment whereas a more dense concentration is restricted to the leaf ornamentation appearing green in tungsten light.
Other types of investigation
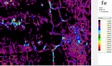
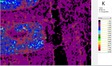
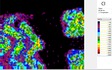
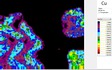
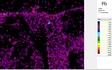
The area of analysis (1 × 1 cm) covers all colours represented on the fragment with the exception of the black paint.
White µ-XRF spectrometry shows a high concentration of Ca which is limited primarily to the white ground between and right above the green leaves (Area 1_Ca). It can be ascribed to calcium carbonate. However, analysis of the white ground beneath the yellow layer provides new and unexpected results. The spectral analysis reveals a high concentration of Pb suggesting the use of lead white as a preparatory layer. Red µ-XRF analysis shows no evidence of the elements Fe, Hg or Pb and the content of As belongs to the yellow colour. This rules out the use of the most common inorganic pigments like ochre and realgar. Also vermillion and red lead pigments not attested before the late Ptolemaic period are unlikely and the red colour remains at present unidentified Green According to the spectral analysis the green colour contains both Cu and Cl which can be attributed to atacamite (Cu₂(OH)₃Cl); a green orthorhombic copper chloride hydroxide mineral used in its synthetic form (Area 1_Cu Area 1_Cl). Even if the intentional use of such basic copper chlorides as green pigments are mentioned in the literature it is notable that they are more frequently attributed to the degradation of other copper-containing pigments, including malachite and green frit. Yellow The µ-XRF reveals a high concentration of S in area 1. Also As is found in high concentrations in the upper half of the fragment and the elemental composition suggests orpiment, a natural arsenic sulphide, AsS₃, with its characteristic bright yellow colour and strong tonality.
Bibliography
C. Brøns, K. Lund Rasmussen, M. Melchiorre di Crescenzo, R. Stacey, A. Lluveras-Tenorio (2018) Painting the Palace of Apries I: Ancient binding media and coatings of the reliefs from the Palace of Apries, Lower Egypt, Heritage Science 6:6 “https://heritagesciencejournal.springeropen.com/articles/10.1186/s40494-018-0170-9”:
S. Hedegaard, T. Delbey, C. Brøns, K. Rasmussen, Painting the Palace of Apries II: ancient pigments of the reliefs from the Palace of Apries, Lower Egypt. Heritage Science. 7 (2019), doi:10.1186/s40494-019-0296-4.
T. Bagh (2011), Finds from W.M.F. Petrie’s Excavations in Egypt in the Ny Carlsberg Glyptotek, in Meddelelser fra NyCarlsberg Glyptotek. No. 13 Copenhagen, 37-43.
M. Jørgensen (2009), Egypt IV Late Egyptian Sculpture 1080 BC-AD 400. Ny Carlsberg Glyptotek, Copenhagen, 91, cat. No. 26.2.
- ÆIN 1058
- Fragment
- 26th dyn. 589-568 B.C.E.
- Egypt: Early Dynastic Period
- Limestone
- W.M.F. Petrie’s excavations at the north end of Memphis unearthed in 1909 remains of the royal palace of Apries. That same year the Glyptotek in Copenhagen received a number of painted limestone relief fragments belonging to the Palace.
- H. 10.5 cm.; W. 8 cm.; D. 2 cm.